Rare earth elements are central to modern technology: smartphones, electric vehicles, and wind turbines all depend on them. Finnish chemist Johan Gadolin identified the first of them, yttrium, in 1794. Their importance became clear only in the mid-20th century, more than 150 years later.1
The family consists of scandium, yttrium, and the 15 lanthanides, which sit between barium and hafnium on the periodic table. Identifying the remaining 16 would take more than a century after Gadolin’s work.2 3
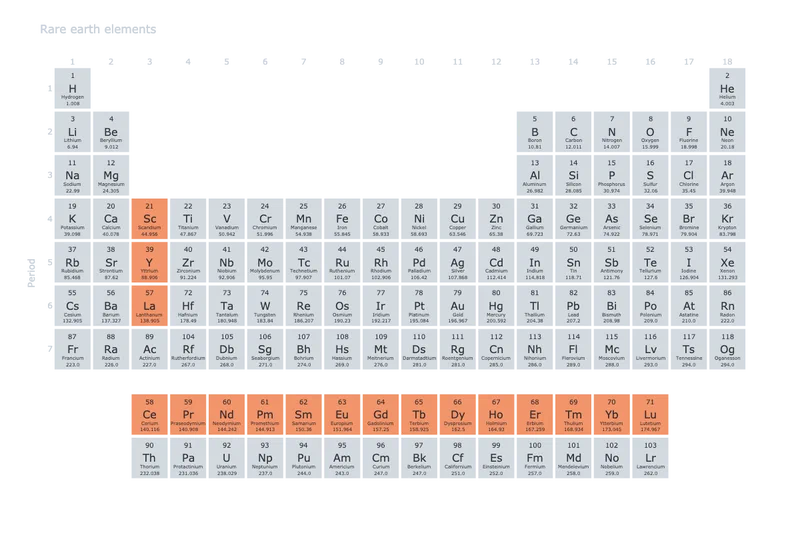
At the time, chemists were identifying new elements at a rapid pace. Rare earths were the most difficult to characterize. Extracted from minerals like yttria and ceria, they did not fit any of the patterns chemists had developed for other elements.
# Neither rare nor earths
The name “rare earths” reflects the assumptions of the chemists who first encountered these substances, not what we now know about them. When chemists first isolated them in the late 1700s, they obtained them as oxides, which were then called “earths” (from the French terre and German Erde).4 The minerals containing them, yttria and ceria, were known only from a single location, the village of Ytterby in Sweden, and their apparent scarcity there gave the family the other half of its name.5 6
Rare earths are not actually rare. They are more abundant in Earth’s crust than mercury or cadmium. The difficulty is that they occur in trace concentrations across many minerals rather than in concentrated deposits, like a thousand euros in cents scattered across a beach instead of a single ten-euro bill in your pocket.
The real difficulty was not scarcity but chemical similarity. The 15 lanthanides have nearly identical electron configurations, which makes separating them like sorting a mix of dark blue and navy blue marbles by color. Specialized techniques are needed to tell them apart.
This chemical similarity also explains why yttria and ceria are not unique sources. The same elements were later found in monazite, a mineral that would become the main industrial source for rare earths. What seemed at first to be a Swedish curiosity is a family of elements distributed throughout Earth’s crust.
# First discoveries
Chemistry advanced rapidly in the late 18th and early 19th centuries. Boyle, Lavoisier, and Dalton established that the elements are the simplest substances, those that cannot be broken down further by chemical means. By the mid-19th century, nearly 70 of the 90 naturally occurring elements had been identified.4
The rare earths were the exception to this rapid progress. The two minerals containing them, yttria and ceria, had been identified by 1787, but the elements within them could not be reliably separated for more than 160 years, and the last of them, promethium, was not isolated until 1947. Even Dmitri Mendeleev and Julius Lothar Meyer, who built the first periodic tables in the 1860s, could not fit them into their classification. The puzzle was only resolved by the development of atomic theory in the 20th century, which showed why the lanthanides in particular have nearly identical chemical properties.
# Yttria
In 1787, Carl Axel Arrhenius, a Swedish army officer and amateur geologist, collected an unusual black mineral near the village of Ytterby and called it simply “black stone.”7 Swedish chemist Bengt Reinhold Geijer published the first report on it in 1788, but the decisive analysis came in 1794, when Finnish chemist Johan Gadolin found that nearly 38% of the stone consisted of an unknown “earth” (now known to be an oxide), along with iron and silicate.
In 1795, Swedish chemist Anders Ekeberg confirmed Gadolin’s findings and named the oxide “yttria” after Ytterby. Like most chemists at the time, Ekeberg treated the new “earth” as an element in its own right, a misconception that persisted until 1808, when Humphry Davy’s electrolysis experiments showed that yttria was a compound. The metallic element within it would later be isolated and named yttrium.
The black stone continued to yield results. In 1802, Ekeberg identified yttria in another mineral, yttrotantalite, and from it isolated tantalum. German chemist Martin Heinrich Klaproth and French chemist Louis Nicolas Vauquelin independently confirmed Gadolin’s analysis. Klaproth then named the original mineral “gadolinite” in his honor.
# Ceria
A second Swedish mineral, cerite, had been suspected of containing an unknown “earth” since the mid-1700s, but confirming it took decades. In 1803, two independent teams reported the result almost simultaneously. The Swedish chemists Jöns Jacob Berzelius and Wilhelm Hisinger were one, and the German chemist Martin Heinrich Klaproth, who had earlier confirmed Gadolin’s work on yttria, was the other. Both teams published their findings in the Neues Allgemeines Journal der Chemie in the same year.
A naming dispute followed. Klaproth proposed “ochroite earth,” while Berzelius and Hisinger proposed “ceria,” after the dwarf planet Ceres, which had been discovered two years earlier. Ceria won, largely due to Hisinger’s standing as an industrialist and patron of science, though most of the analytical work had likely been done by the young Berzelius.
The two teams interpreted the result differently. Klaproth treated the new “earth” as an element in its own right, the same misconception that had attached to yttria. Berzelius correctly identified it as the oxide of a new element, an interpretation consistent with the work that would later make him one of the founders of modern chemistry: precise atomic mass calculations, the formalization of atomic theory, and the modern system of chemical symbols. Klaproth, already known for the discovery of uranium and zirconium, continued his work on strontium, titanium, and tellurium.
The yttria and ceria discoveries set a pattern that would repeat throughout the 19th century: the rare earths rarely came alone, and characterizing them required collaboration across national boundaries.
# Mosander’s investigation
It took two decades for chemists to realize that the original “earths” were not pure elements but metal oxides. Ceria became cerium oxide, yttria became yttrium oxide. The reclassification was straightforward, but the trouble began with the atomic masses.
Measurements of the atomic masses of these supposedly pure elements varied between samples, which had no good explanation if the elements really were pure. The most plausible interpretation was that they were mixtures.
Carl Gustaf Mosander, assistant to Jöns Jacob Berzelius, took up the problem in the 1820s and worked on it for the next two decades. He began with cerium, subjecting it to a long series of reactions. In one experiment he heated cerium oxide with sodium and chlorine to obtain metallic cerium, and he noticed that cerium compounds varied in color and density depending on their mineral source. He concluded that cerium oxide must contain at least one additional element. His colleague Friedrich Wöhler urged him to publish, but Mosander refused to commit to a result until he was certain of it, a caution that frustrated his colleagues but was essential to the discoveries that followed.
# Two earths become six
In 1839, after years of work, Mosander confirmed that cerium oxide was not a single substance. He isolated a new element from it and named it lanthanum, from the Greek for “to escape notice.”8 Two years later, in 1841, he identified a third element in cerium oxide and named it didymium, from the Greek for “twin,” because its chemistry resembled that of lanthanum. Wöhler and Berzelius found the name unusual, but the result stood. Didymium would itself turn out to be a mixture in the 1880s, when it was resolved into praseodymium and neodymium.
Mosander’s analytical methods were as important as the elements he isolated. He developed techniques for repeated crystallization and gravimetric analysis that required extreme care, and in 1843 Berzelius documented them in detail, showing how Mosander had separated cerium, lanthanum, and didymium from what had appeared to be a single substance.
Mosander then applied the same methods to yttrium oxide, working from gadolinite. In 1843 he isolated two more elements, erbium and terbium, with yttrium as the remaining fraction. He used oxalate precipitation to separate them, exploiting differences in reactivity and in the color and crystal form of the precipitates. What had begun as two “earths,” cerium and yttrium, was now six elements, and the rare earths were not what chemists had first taken them to be.
# Spectral analysis
Mosander’s separation methods were slow. A single result could require months or years of repeated crystallizations and precise measurements, and as organic chemistry advanced through the middle of the 19th century, work on the rare earths slowed.
In 1859, Robert Bunsen and Gustav Kirchhoff introduced spectral analysis. Heated elements emit light at characteristic wavelengths, and the resulting pattern of spectral lines is unique to each element. Identification could now be made directly from the spectrum, without the long sequence of separation steps that Mosander had relied on.
The technique arrived just as Dmitri Mendeleev and Julius Lothar Meyer were independently developing the periodic table in the late 1860s. Their table organized the elements by atomic weight and chemical properties, exposing relationships between them and even predicting the existence of elements that had not yet been found.
The two tools were complementary. The periodic table identified gaps where new elements should exist, and spectral analysis allowed chemists to look for them directly. The slow crystallization work of Mosander’s generation gave way to a faster mode of discovery.
# A new generation
Spectral analysis and the periodic table brought a new generation of chemists to the rare earths. Swiss chemist Jean Charles Galissard de Marignac built on Mosander’s methods to recalculate the atomic weights of cerium, lanthanum, and didymium with much greater precision. His measurements arrived shortly after the 1860 Karlsruhe Congress, where the need for standardized atomic weights had been the central question, and they helped European chemists converge on consistent values.
Another Swiss chemist, Marc Delafontaine, worked on Mosander’s discoveries, in particular terbium. Several chemists had worked on isolating it, which makes the discovery hard to attribute to a single individual, but Delafontaine’s systematic studies clarified its properties. He then turned to didymium, the “twin” element Mosander had identified, and noticed subtle differences in its spectral lines that suggested it was not a single element.
The French chemist Paul Émile Lecoq de Boisbaudran later confirmed this. Working with samarskite, a mineral rich in rare earths, he used spectral analysis to show that didymium was a mixture. It would eventually be resolved into praseodymium and neodymium. The pattern set by Mosander’s lanthanum and his original didymium repeated itself: an element that had been treated as single turned out to be several.
# The final discoveries
In 1885, Austrian chemist Carl Auer von Welsbach succeeded where others had failed and separated didymium into two elements. One gave green oxides, which he named praseodidymium (“green twin”), and the other gave pink oxides, which he named neodidymium (“new twin”). They are now known as praseodymium and neodymium.
Von Welsbach pursued the practical applications as well as the chemistry. His incandescent gas mantle, which used rare-earth salts, mainly thorium and cerium, to produce bright light from a gas flame, was the basis of the lighting company Osram and made him commercially successful. It is one of the earliest cases of rare-earth research moving directly into industrial use.
In 1896, Eugène-Anatole Demarçay identified europium in samarium samples and named it after Europe. At the end of the 19th century, von Welsbach and the French chemist Georges Urbain independently showed that ytterbium contained two elements. The names they proposed (Urbain’s neoytterbium and lutetium, von Welsbach’s aldebaranium and cassiopeium) gave rise to a dispute that lasted for decades. In Germany, “cassiopeium” remained in use in place of lutetium until the Second World War.
Element 61 was the last gap. It was filled in 1947, when scientists at the Manhattan Project isolated traces of it from uranium fission products and named it promethium, after the Titan who brought fire to humanity. Promethium is highly radioactive and occurs in nature only in trace amounts.
With promethium, the family was complete. What had begun in the 1780s with two unidentified “earths” had become a chemical family of 17 elements. The history of their discovery is less a story of breakthroughs than of patient, generational work: a small number of chemists, working in close communication across national borders, slowly resolved a chemistry that had no good analytical handle until the 20th century gave them one.
Critical Metals Handbook, 1st ed.; Gunn, G., Ed.; John Wiley & Sons, 2013. https://doi.org/10.1002/9781118755341 . ↩︎
Höppe, H. Rare-Earth Elements: Solid State Materials: Chemical, Optical and Magnetic Properties; De Gruyter Graduate; De Gruyter: Berlin, DE, 2024. https://doi.org/10.1515/9783110680829 . ↩︎
Voncken, J. H. L. The Rare Earth Elements; SpringerBriefs in Earth Sciences; Springer: Cham, CH, 2016. https://doi.org/10.1007/978-3-319-26809-5 . ↩︎
Gschneidner, K. A.; Eyring, L. Handbook on the Physics and Chemistry of Rare Earths: Two-Hundred-Year Impact of Rare Earths on Science; Handbook on the Physics and Chemistry of Rare Earths; Elsevier Science: Amsterdam, NL, 1988; Vol. 11. ↩︎ ↩︎
Rowlatt, J. Rare Earths: Neither Rare, nor Earths. BBC News. March 23, 2014. https://www.bbc.com/news/magazine-26687605 (accessed 2024-08-18). ↩︎
Klinger, J. M. A Historical Geography of Rare Earth Elements: From Discovery to the Atomic Age. Extr. Ind. Soc. 2015, 2 (3), 572–580. https://doi.org/10.1016/j.exis.2015.05.006 . ↩︎
Not to be confused with Svante Arrhenius, the Swedish Nobel Prize winner who was born in 1859. ↩︎
Episodes from the History of the Rare Earth Elements; Evans, C. H., Ed.; Springer Netherlands: Dordrecht, NL, 1996. https://doi.org/10.1007/978-94-009-0287-9 . ↩︎