Glass is in windows, containers, optical fibers, lenses, and the substrates of most electronic displays. Its atomic structure differs from that of most solids: in crystals, atoms are arranged in a periodic lattice, but in glass they are not.1 This disorder is the source of much of what is distinctive about glass, including its gradual softening with temperature instead of a sharp melting point and its isotropic mechanical response. Glass is also out of thermodynamic equilibrium: its structure is slowly relaxing toward a more stable arrangement, but the process is too slow to observe at ordinary temperatures.
# Crystalline and amorphous solids
Solids fall into two structural categories: crystalline and amorphous. In crystalline solids, such as diamond or metallic copper, atoms occupy a highly ordered, repeating lattice. In amorphous solids, including glass, the atoms are disordered, resembling a liquid frozen in place rather than a regular lattice.2 3
The periodic arrangement gives crystalline solids two characteristic properties. They have a sharp melting point: at the right temperature, the entire lattice breaks down at once. They are also anisotropic, meaning their mechanical properties depend on the direction of an applied force, because some directions in the lattice are stronger than others.
Glass differs in both respects. Because there is no lattice to break, it does not have a sharp melting point and softens gradually as temperature rises, transitioning from rigid to fluid over a range. The disordered atomic arrangement has no preferred direction, so glass is isotropic: its mechanical properties are the same along every axis.
The contrast is sharpest in silica (). In its crystalline form, silica is quartz, with a sharp melting point and anisotropic mechanical properties. In its amorphous form, the same chemical composition produces a glass with no melting point and isotropic properties.
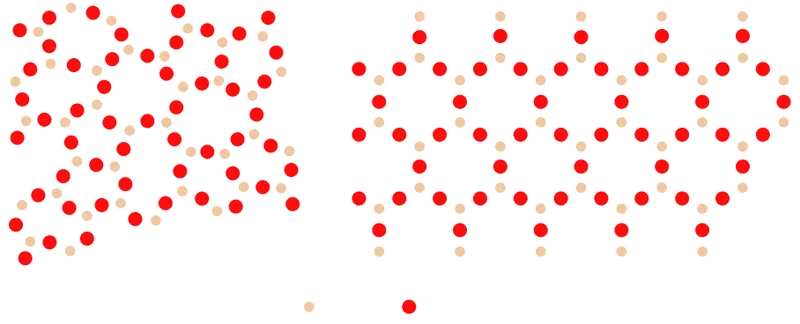
Glass is not limited to silica. Many other substances, including metals and polymers, can form amorphous structures when cooled rapidly enough to suppress crystallization. What they all share is the absence of long-range order in the atomic arrangement.
# Metastability
Glass is metastable: it sits in a local energy minimum that is not the global minimum, but the energy barrier separating the two is too large to cross under ordinary conditions. For almost any glass-forming substance, the global minimum is a crystalline arrangement.4 5
Crystallization requires atoms to settle into the periodic positions of a lattice, and that takes time. When a melt is cooled fast enough, there is not enough time for them to do so, and the resulting solid carries the disordered arrangement it had in the liquid state. The faster the cooling, the more frozen-in disorder.
Compared to a crystal of the same composition, a glass has a larger molar volume (its atoms are not packed as tightly), higher entropy (the disordered arrangement has more accessible microstates), and higher enthalpy and Gibbs free energy. All of these reflect the same fact: glass is in a higher-energy state, but the activation energy required to reach the lower-energy crystalline state is too large to cross at ordinary temperatures.
# The glass transition
The transformation from a glass-forming liquid into a glass is called the glass transition, and the process of carrying it out (cooling a liquid fast enough to bypass crystallization) is called vitrification. Unlike the melting transition of a crystal, which happens at a single sharp temperature, the glass transition is gradual: as a liquid is cooled, its viscosity rises, and over some temperature range it becomes so viscous that, on the timescales of observation, it stops flowing. The temperature at which this happens is called the glass transition temperature, .3
is not a thermodynamic constant of the material but a quantity that depends on the cooling rate. Faster cooling traps the atoms at a higher temperature, before they have time to relax into lower-energy arrangements, and produces a higher . Slower cooling gives them more time to relax and produces a lower . The same substance can have different glass transition temperatures depending on its thermal history.
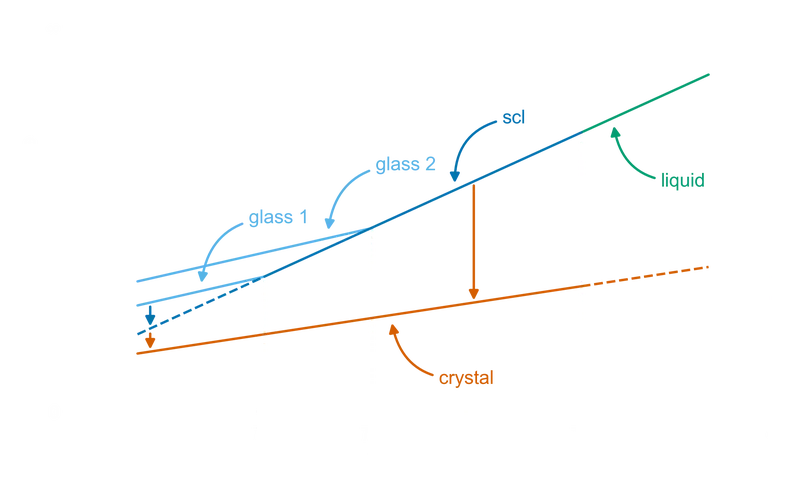
This time-dependence is the central distinction between glass and crystalline solids. Crystals melt at a fixed temperature (), and the transition is sharp. Glasses do not melt at all in the strict sense. They pass continuously between rigid solid and viscous liquid, and the location of the transition depends on time as much as on temperature.
# Supercooled liquids
A liquid cooled below its freezing point without crystallizing is a supercooled liquid. As it cools further, its atomic structure continues to rearrange through a process called structural relaxation, which slows as the temperature drops. The characteristic timescale for this rearrangement is the relaxation time, , which increases sharply as the liquid approaches the glass transition.
When the cooling rate exceeds the structural relaxation rate, the atomic arrangement is frozen in: continued cooling produces no further structural change, and the supercooled liquid becomes a glass. The temperature at which this happens is called the fictive temperature, and for many materials it coincides with .
# Conclusion
Glass is structurally disordered, thermodynamically out of equilibrium, and stable on human timescales. These features, together with the glass transition that connects the liquid and solid states, are common to all glasses, whether oxide, metallic, or polymeric. What varies between systems is the chemistry that sets the relaxation timescales and the value of .
Mysen, B.; Richet, P. Silicate Glasses and Melts, 2nd ed.; Elsevier: Amsterdam, NL, 2019. https://doi.org/10.1016/C2018-0-00864-6 . ↩︎
Vogel, W. Glass Chemistry; Springer: Berlin, DE, 1994. https://doi.org/10.1007/978-3-642-78723-2 . ↩︎
Schmelzer, J. W. P.; Gutzow, I. S. Glasses and the Glass Transition; John Wiley & Sons: Weinheim, DE, 2011. https://doi.org/10.1002/9783527636532 . ↩︎ ↩︎
Zanotto, E. D.; Mauro, J. C. The Glassy State of Matter: Its Definition and Ultimate Fate. J. Non-Cryst. Solids 2017, 471, 490—495. https://doi.org/10.1016/j.jnoncrysol.2017.05.019 . ↩︎
Montazerian, M.; Zanotto, E. D. The Glassy State. In Encyclopedia of Materials: Technical Ceramics and Glasses; Elsevier, 2021; Vol. 2, pp 448—461. https://doi.org/10.1016/B978-0-12-803581-8.11728-X . ↩︎